The £1 Pill That Could Transform Cancer Care
How low-dose aspirin halves colorectal cancer recurrence for patients with key genetic mutations
Why it matters
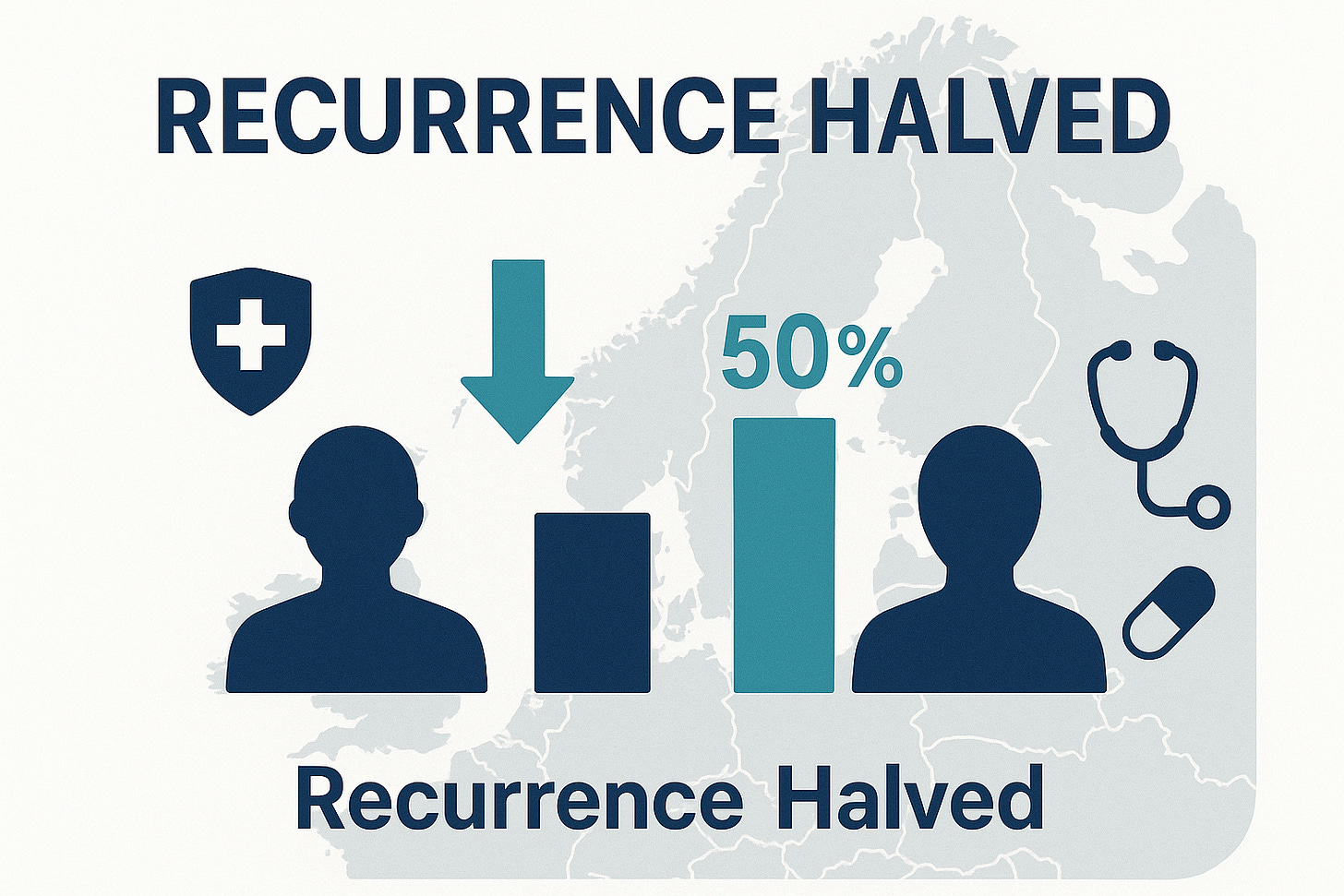
A single daily tablet costing less than 10p has delivered one of the most remarkable breakthroughs in modern oncology. The ALASCCA trial, published in the New England Journal of Medicine in September 2025, shows that low-dose aspirin can cut the recurrence of colorectal cancer by around 50 per cent in patients carrying specific PI3K-pathway mutations.
This discovery fuses 20th-century pharmacology with 21st-century genetics—a simple, globally available medicine tailored by precision DNA testing.
A Scandinavian study changes the rules
The ALASCCA (Adjuvant Low-Dose Aspirin in Colorectal Cancer) trial enrolled 3,508 patients across 33 hospitals in Sweden, Norway and Denmark. Every participant had undergone curative surgery for localised colorectal cancer. Genetic sequencing then sorted them by mutations in the PI3K signalling pathway—a key cellular circuit controlling growth and survival.
Roughly 37 per cent of screened patients showed alterations in this pathway, mainly in the PIK3CA, PIK3R1 or PTEN genes. They were randomly assigned to receive either daily low-dose aspirin (160 mg) or placebo for three years.
The results were emphatic: recurrence rates halved.
Group A (hot-spot PIK3CA mutations): 14.1 % → 7.7 % (51 % risk reduction).
Group B (other PI3K alterations): 16.8 % → 7.7 % (58 % reduction).
Overall disease-free survival climbed from 81–79 % in the placebo arms to almost 89 % with aspirin.
The biology behind the benefit
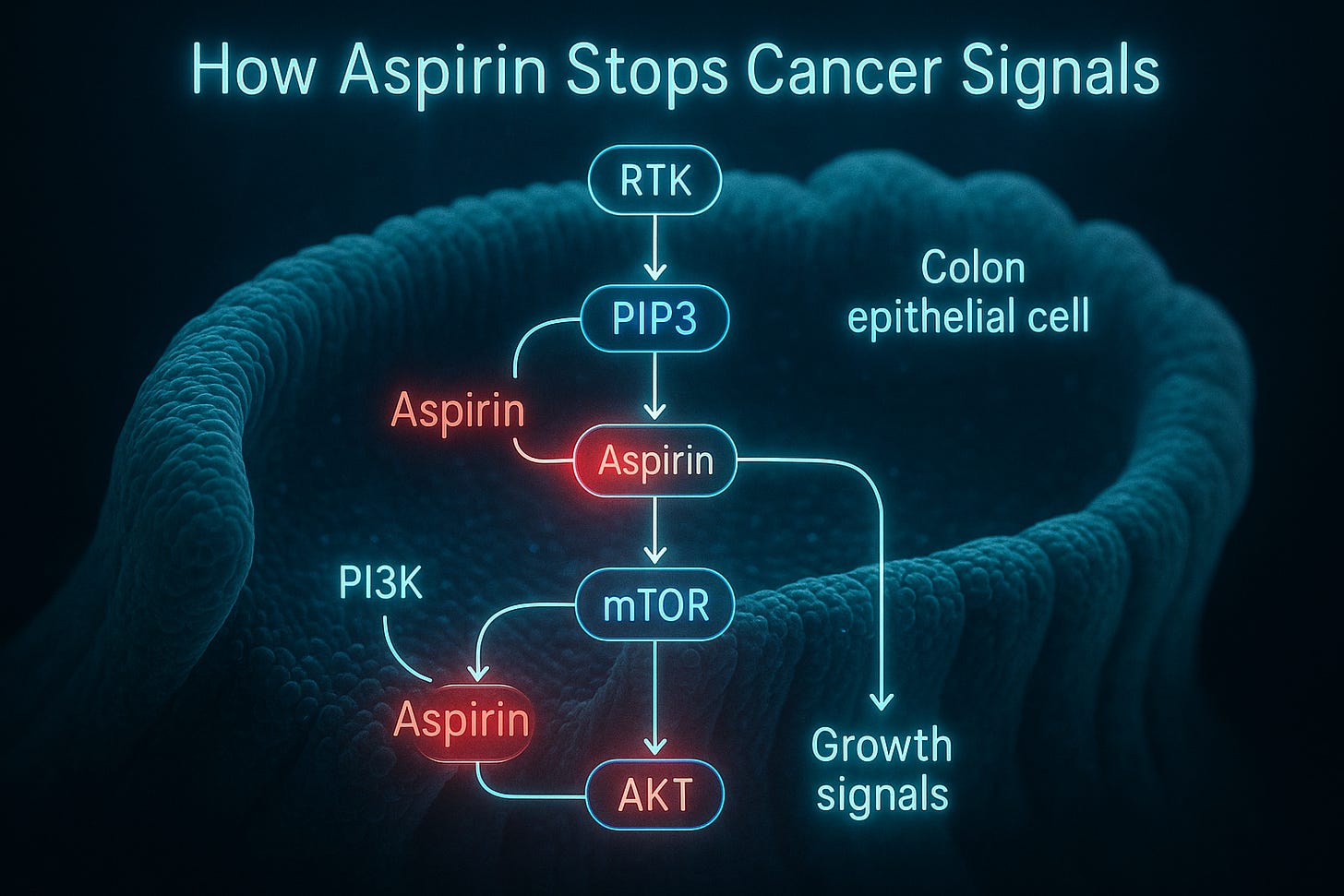
Aspirin’s anti-cancer power is no fluke. It suppresses cyclo-oxygenase enzymes (COX-1 and COX-2), damping chronic inflammation and platelet activation—two forces known to aid metastasis [9][10]. But in PIK3CA-mutated tumours, the effect appears magnified because the same pathway controls both inflammation and cell proliferation.
In essence, aspirin shuts the fuel valve on a mutation-driven growth engine. By interrupting multiple signals at once—COX, platelets, and the PI3K cascade—it deprives malignant cells of their micro-environmental support.
Affordable precision medicine
Professor Anna Martling of Karolinska Institutet called the findings “a clear example of how we can use genetic information to personalise treatment and at the same time save both resources and suffering”.
Unlike many targeted therapies priced in tens of thousands of pounds per course, aspirin costs pennies a day and is already stocked in every pharmacy on Earth. For health systems under financial strain, it could become a model of equitable precision care—a low-tech, high-impact intervention guided by genetic testing.
Economic analyses confirm the attraction. Aspirin chemoprevention consistently falls well below the UK cost-effectiveness threshold of £50,000 per QALY. When limited to the one-third of patients most likely to respond, its value improves even further.
From trial to guideline
The ripple effect was immediate. The National Comprehensive Cancer Network (NCCN) updated its 2025 colon-cancer guidelines to recommend PIK3CA testing for stage II–III disease. For patients with qualifying mutations, daily low-dose aspirin (100–162 mg) is now endorsed as adjuvant therapy.
It is the first time a 19th-century compound has been written into a modern precision-oncology protocol.
Beyond the gene: a systems view of cancer
Traditional oncology focused on a few well-known biomarkers such as KRAS or BRAF. ALASCCA pushes the frontier further, treating the PI3K network itself as a therapeutic target.
This shift marks a conceptual evolution:
From disease type to molecular profile.
From population averages to individual predictors.
From expensive novel drugs to intelligent reuse of existing ones.
Aspirin’s impact may also involve the gut microbiome. Early studies suggest it encourages beneficial bacterial species that reinforce the intestinal barrier and limit tumour inflammation. Precision medicine could soon mean sequencing both tumour DNA and gut flora before writing a prescription.
Balancing risk and reward
No medicine is without downside. In ALASCCA, 16.8 per cent of aspirin users experienced severe adverse events versus 11.6 per cent on placebo. Most were gastrointestinal bleeds.
Yet when weighed against the halving of cancer recurrence, the net clinical benefit remains compelling. The 160 mg dose chosen by investigators strikes a middle ground—strong enough for anti-tumour effects but below the anti-inflammatory range linked to higher bleeding risk.
Implementation in real-world practice will require:
careful patient selection (exclude high-risk bleeders);
proton-pump inhibitor co-therapy where appropriate;
routine genetic testing to verify PI3K-pathway status.
Education and dissemination
Clinicians can explore detailed analysis in Dr Mobeen Syed’s explainer Aspirin Reduces the Risk of Colorectal Cancer [Link] and Professor Karen Brown’s lecture Bowel Cancer Prevention – Is Aspirin Just the Start? [Link]. For a grounding in precision medicine principles, the FDA Oncology Center’s video What Is Precision Medicine? offers a concise introduction.
These resources will be crucial as oncology teams integrate genetic screening into post-surgical follow-up and as policymakers weigh national roll-outs of testing.
The bigger horizon: a platform discovery
Aspirin’s PI3K-dependent mechanism may extend beyond the bowel. PIK3CA mutations are common in breast, endometrial and prostate cancers. Researchers are already planning cross-tumour trials to test whether low-dose aspirin provides similar recurrence reductions in these settings.
If confirmed, oncology could gain its first multi-cancer precision adjuvant—a single agent applicable wherever the same molecular wiring is present.
The implications for global health equity are profound. A therapy costing £30 a year could lower relapse rates in millions of patients, particularly in regions where access to biologics and immunotherapy remains limited.
Towards the clinic of the future
The ALASCCA findings illustrate what the next decade of medicine might look like:
Genome profiling as routine. A short DNA panel identifies key pathway alterations.
Drug repurposing by design. Existing medicines are matched to molecular signatures.
Global scalability. Low-cost generics enable immediate adoption across income levels.
Continuous data feedback. Real-world outcomes feed machine-learning models to refine eligibility.
This fusion of data science and pharmacology represents the maturing of precision medicine—from theory to public-health instrument.
Caveats and next questions
Regulators will want to see replication in non-Scandinavian populations and longer follow-up for late relapse. Questions also remain over optimal duration (is three years enough?) and combination therapy (could aspirin complement immunotherapy?).
There is also an ethical debate over genetic testing logistics—who pays, who owns the data, and how to ensure equitable access. The UK’s Genomics Medicine Service could become a testbed for integrating such low-cost pharmacogenomic interventions into the NHS cancer pathway.
Conclusion: science returns to simplicity
For decades, progress in cancer therapy has often meant ever more complex and expensive molecules. The ALASCCA trial turns that logic inside-out. Sometimes innovation lies not in invention but in re-interpretation—finding new purpose in old drugs through the lens of molecular data.
Aspirin, first synthesised in 1897, may now anchor a new chapter of precision oncology. Its success proves that personalisation and affordability need not be opposites.
If adopted globally, this humble tablet could prevent tens of thousands of recurrences each year, save billions in treatment costs, and—more importantly—give survivors the quiet miracle of an ordinary life resumed.